Synergy 2000

- Enterprise-wide monitoring
- Enhanced regulatory standards with powerful gage R & R functionality
- Rapid set-up and implementation for quick ROI
- Flexible data entry methods
- Easy, one-click navigation
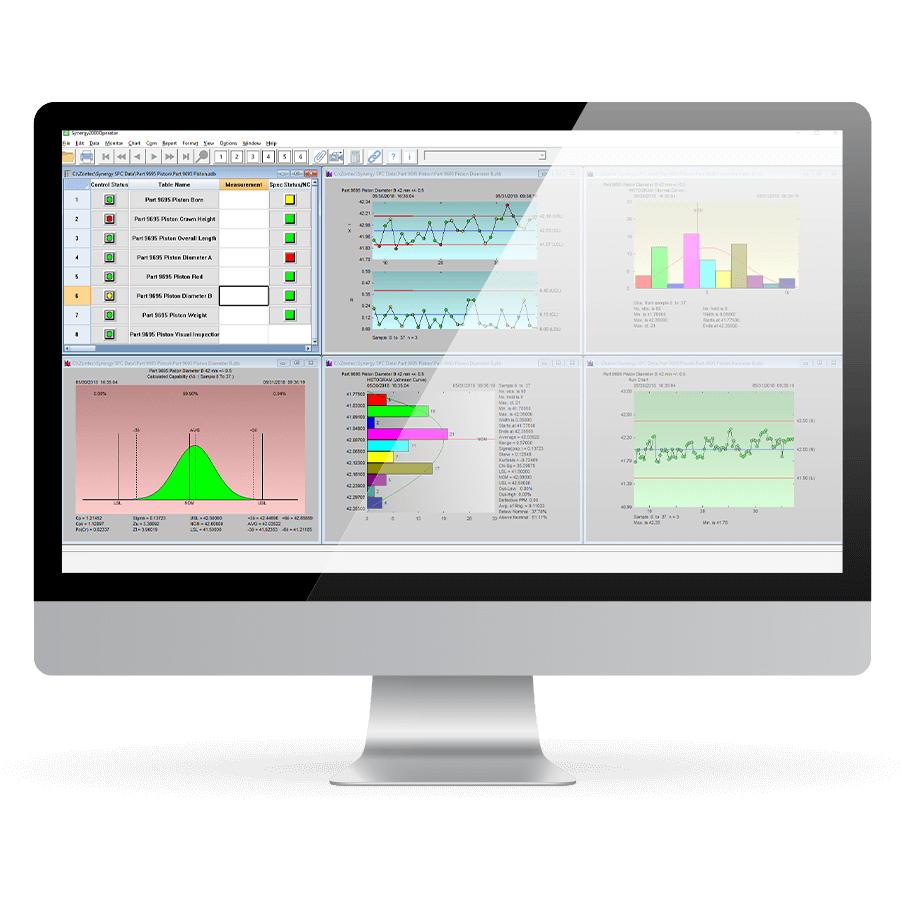
- Wide variety of interactive charts
- Sophisticated analytics and real-time math calculations
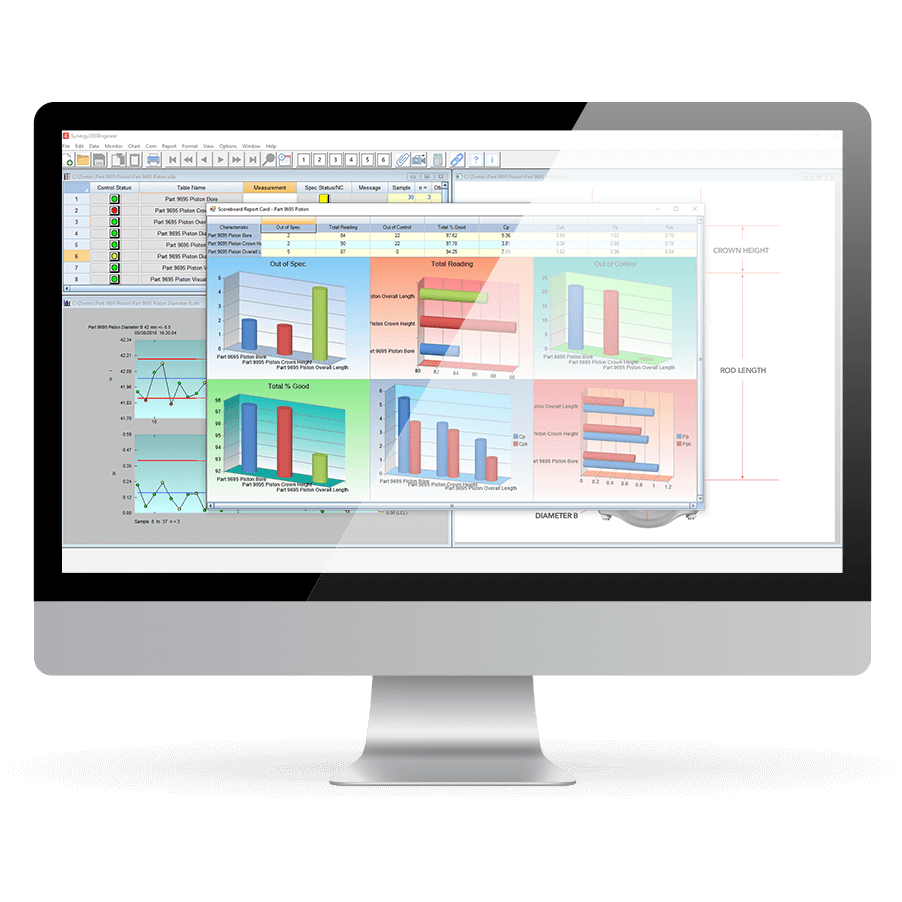
- Instant extensive reporting
- Automated data collection with direct input devices
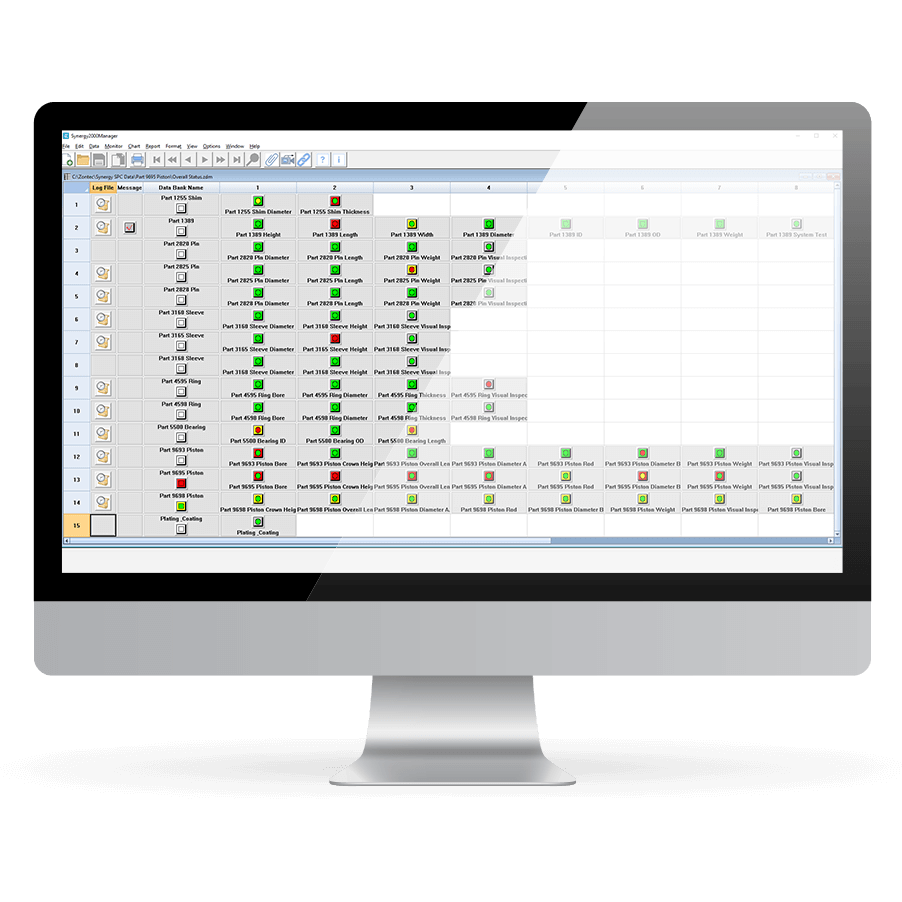
- Stringent access and security controls
- 1 GHz or faster PC processor
- 1.0GB of RAM minimum
- Microsoft Windows 7 or greater
- 1024x768 or higher resolution monitor
- 1 GHz server (or better) with at least 10GB hard drive
- Windows Server 2008 or greater
- Microsoft .NET 4.0 or greater
- Optional: Automated data collection devices
Monitor Quality in Real-Time, Enterprise-Wide
Zontec gives you instant collaboration so your manufacturing team can make quality a proactive part of your business. Rich in features and functionality, our Synergy SPC software serves over 5000 companies in more than 50 countries.
Simple, Fast Operation
Easy to navigate screens make data collection, charting, monitoring, analysis, communication, and reporting simple and straightforward. And you can do it all with the click of a mouse.

Wide Variety of Reports
Synergy 2000 offers superior reporting, including performance metrics, profitability analysis, and reports such as the Automated Certificate of Analysis to meet customer requirements.

Instant, Enterprise-Wide Communication
Synergy 2000 is designed so all information can be viewed by or emailed to the appropriate person wherever they are in the enterprise instantly.

Enhanced Security Controls
Synergy 2000 provides the most stringent access and security controls for quality and safety data. It satisfies the needs of the U.S. Food and Drug Administration for full compliance with Title 21 Code of Federal Regulations (CFR) Part 11.

Case Study
The Carlisle & Finch Co Uses Synergy to Reduce Its Defects to Customers to Zero
Founded in 1894, The Carlisle & Finch Co. invented the first Carbon Arc Searchlight in North America, and provided this technology to the Paddlewheel Boats on the Ohio and Mississippi Rivers. In the 1960’s, Carlisle & Finch pioneered Xenon Arc Searchlight Technology, and the Company’s products remain to this day, the standard of the U.S. Navy, the U.S. Coast Guard and the U.S. Federal Prison System.

50%
Reduced Part Inspection

2/3rds
Reduced Rejects

“I think that where Synergy has helped our company the most has been on final inspections. Since implementing Synergy we have reduced our rejects by 2/3rds.”
—Bob Batsche, Quality Manager for mf